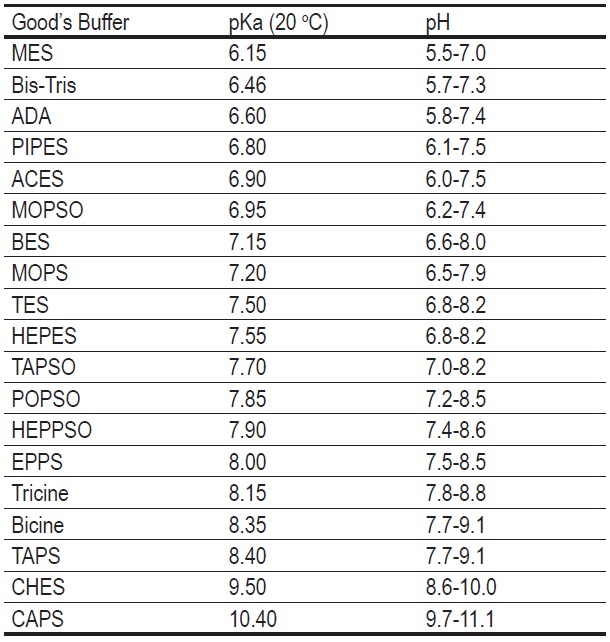
Buffer solutions in drug formulation and processing: How pKa values depend on temperature, pressure and ionic strength - ScienceDirect

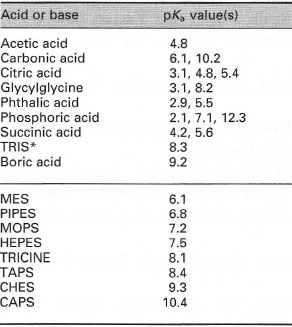
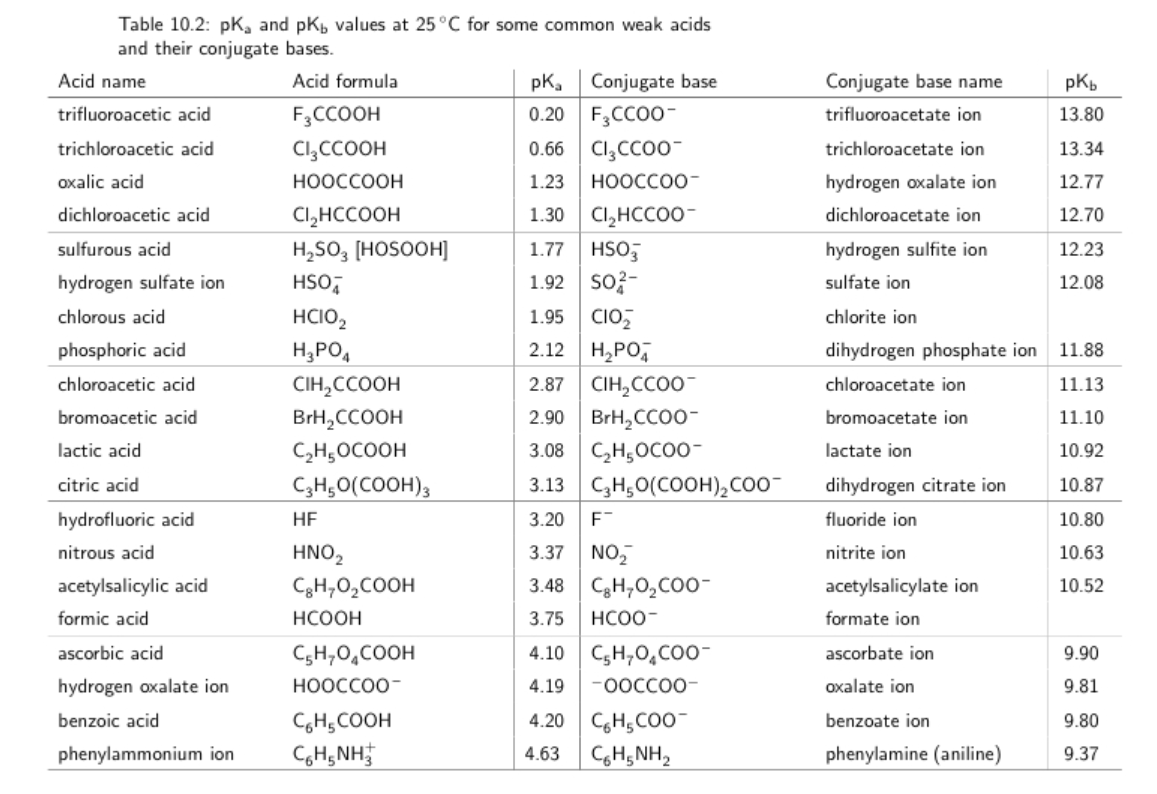
SOLVED: 11. Buffer preparation Given the KalpKa of the following compounds: TABLE 8.3 Some K; and pK; Values for Weak Acids, in Decreasing Order of Strength Name Formula K pK. Phosphorie acid
OneClass: A buffer solution is prepared by mixing 43.1 mL of 0.0478 M sodium dihydrogen citrate with ...

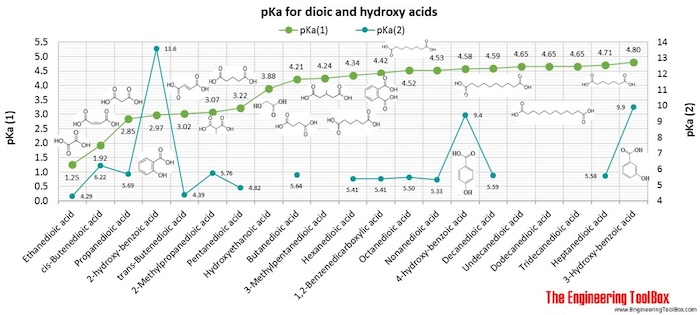
A Reliable and Efficient First Principles-Based Method for Predicting pKa Values. 2. Organic Acids | The Journal of Physical Chemistry A

Table 4 from Measurement of dissociation constants (pKa values) of organic compounds by multiplexed capillary electrophoresis using aqueous and cosolvent buffers. | Semantic Scholar
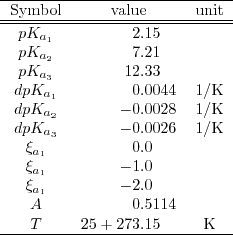
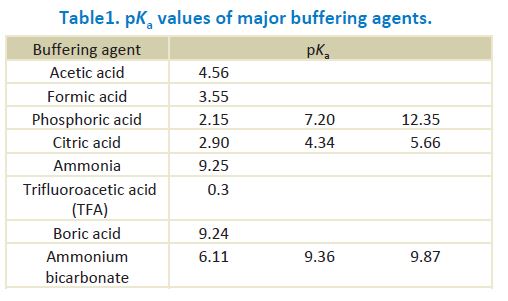
![PDF] The Influence of Ionic Strength on Apparent and Thermodynamic Parameters ( Ka , pKa ' s ) for HF and Phosphate Buffer Capacities | Semantic Scholar PDF] The Influence of Ionic Strength on Apparent and Thermodynamic Parameters ( Ka , pKa ' s ) for HF and Phosphate Buffer Capacities | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/c0a6140f88db06398f56b28ce5fd6ac92cd82d39/5-Table3-1.png)
PDF] The Influence of Ionic Strength on Apparent and Thermodynamic Parameters ( Ka , pKa ' s ) for HF and Phosphate Buffer Capacities | Semantic Scholar

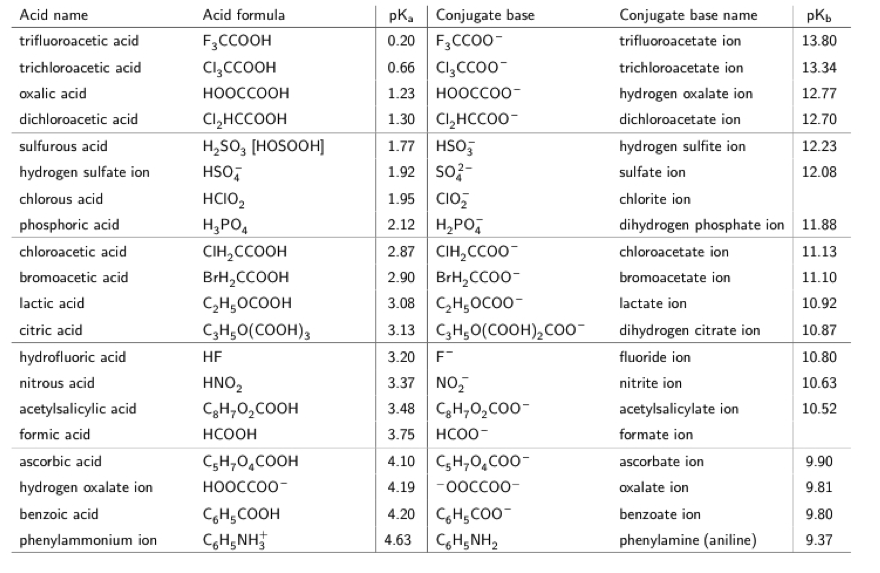
SOLVED: You are trying to create a buffer with pH = 7.7. a.) Which acid from the table below should you use? b.) What else do you need to add to create

Solution pKa values for a range of polymers at various polymer and salt... | Download Scientific Diagram
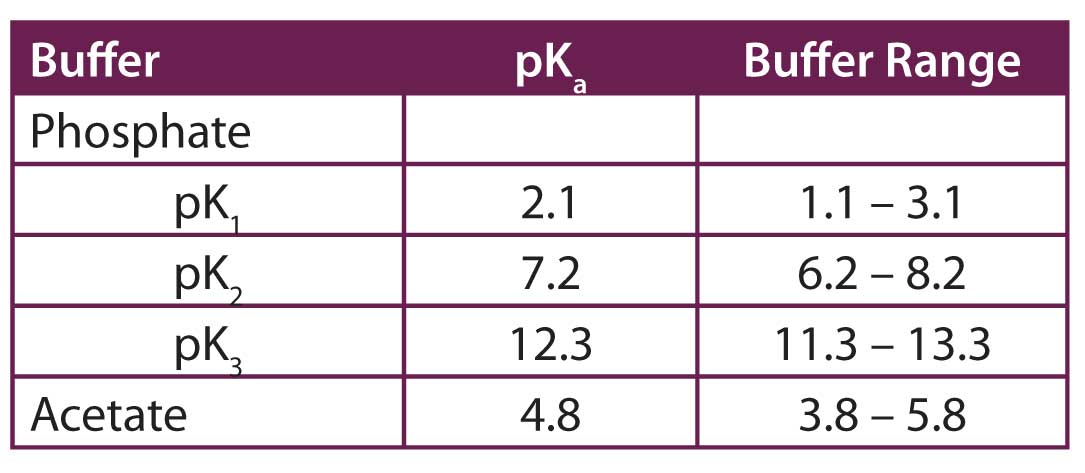
![PDF] Buffer solutions in drug formulation and processing: How pKa values depend on temperature, pressure and ionic strength | Semantic Scholar PDF] Buffer solutions in drug formulation and processing: How pKa values depend on temperature, pressure and ionic strength | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/879d3eae13956041f4cc1b8ee223109275087b7e/3-Table1-1.png)
PDF] Buffer solutions in drug formulation and processing: How pKa values depend on temperature, pressure and ionic strength | Semantic Scholar

Table 3 from Measurement of dissociation constants (pKa values) of organic compounds by multiplexed capillary electrophoresis using aqueous and cosolvent buffers. | Semantic Scholar